

Loss of Muscle Mass Induced by Aging
Pérdida de masa muscular inducida por el envejecimiento
Perda de massa muscular induzida por envelhecimento
Loss of Muscle Mass Induced by Aging
Revista Ciencias de la Salud, vol. 17, no. 2, 2019
Universidad del Rosario
Received: 20 February 2019
Accepted: 18 March 2019
Additional information
To cite this
article: Pinzón-Ríos ID. Loss of Muscle Mass Induced by Aging. Rev Cienc Salud. 2019;17(2):223-44. Doi: https://doi.org/10.12804/revistas.urosario.edu.co/revsalud/a.7925
Abstract: Introduction: The world population ages rapidly, understanding the anatomo-functional changes of the muscle allows the implementation of effective actions in this age group. The objective of this work was to identify, through a review of scientific literature, the current advances in the molecular processes related to the loss of muscle mass induced by aging. Materials and Methods: The molecular causes of muscle mass loss induced by aging were reviewed through scientific articles from 2006 to 2016 on the Pubmed, Scielo and PEDro databases, combining the terms muscular aging, exercise and physical therapy. Results: Three sections were determined: 1) sarcopenia and aging, 2) molecular pathways of muscle mass loss induced by aging and 3) aging, exercise and functionality. Conclusion: Knowing the molecular basis of muscle mass loss induced by aging allows the physiotherapist to understand, evaluate and intervene related mechanisms to perform actions that improve the functionality and quality of life of older adults.
Keywords Aging, exercise, physical therapy.
Resumen: Introducción: la población mundial envejece rápidamente y comprender los cambios anatomo-funcionales del músculo permite implementar acciones eficaces en este grupo etario. El objetivo de este trabajo fue identificar, por medio de una revisión de literatura científica, los avances actuales de los procesos moleculares relacionados con la pérdida de masa muscular inducida por el envejecimiento. Materiales y métodos: se revisaron las causas moleculares de la pérdida de masa muscular inducida por el envejecimiento, mediante artículos científicos publicados desde 2006 hasta 2016 en las bases Pubmed, Scielo y PEDro, combinando los términos: muscular aging, exercise y physical therapy. Resultados: se determinaron tres apartados: 1) sarcopenia y envejecimiento, 2) vías moleculares de la pérdida de masa muscular inducida por el envejecimiento y 3) envejecimiento, ejercicio y funcionalidad. Conclusión: conocer las bases moleculares de la pérdida de masa muscular inducida por el envejecimiento permite al fisioterapeuta entender, evaluar e intervenir los mecanismos relacionados para realizar acciones que mejoren la funcionalidad y calidad de vida de los adultos mayores.
Palabras clave: envejecimiento, ejercicio, fisioterapia.
Resumo: Introdução: a população mundial envelhece rapidamente e compreender as mudanças anátomo-funcionais do músculo, permite implementar ações eficazes neste grupo etário. O objetivo deste trabalho foi identificar através de uma revisão de literatura científica, os avanços atuais dos processos moleculares relacionado com a perda de massa muscular induzida pelo envelhecimento. Materiais e métodos: se revisaram as causas moleculares da perda de massa muscular induzida pelo envelhecimento, mediante artigos cientpificos desde 2006 até 2016 nas bases Pubmed, Scielo e PEDro, combinando os termos: muscular aging, exercise e physical therapy. Resultados: determinaram-se três apartados: 1) Sarcopenia e envelhecimento, 2) Vias moleculares da perda de massa muscular induzida pelo envelhecimento, e 3) Envelhecimento, exercício e funcionalidade. Conclusão: conhecer as bases moleculares da perda de massa muscular induzida pelo envelhecimento, permite ao fisioterapeuta compreender, avaliar e intervir os mecanismos relacionados para realizar ações que melhorem a funcionalidade e qualidade de vida dos adultos maiores.
Palavras-chave: envelhecimento, exercício, fisioterapia.
Introduction
In recent years, decoding the genetic code and the sequencing the human genoma have enabled the emergence of methods from the molecular biology in order to deepen knowledge of the structure and functioning of human tissues from genesis to aging (1). World population is ageing fast, understanding the associated morphologic, physiologic, psychologic and functional changes is a relevant aim for copying with the biomedical and social demands of this age group. One of the alterations that affect quality of life (ql) is the loss of the muscle tissue induced by aging (muscle atrophy) with its subsequent loss of strength (called sarcopenia –sp–), present by the age of 55 years old, when the loss of muscle mass can reach older ages, with highly critical muscle losses (2).
In order to understand the molecular mechanisms responsible of the muscle atrophy, it would be started by the meaning that derives from the Greek atrophos that expresses “without nutrition”, which is a very organized and regulated process that ends in the drop of the muscle protein content reflected in the decrease of the area of the transverse muscle, regardless the possible causes that can lead to this condition (3). Usually, in the clinical care attention is payed to the muscle in different physical capacities, but sometimes it is not dimensioned what the muscle mass for the general health means. Therefore, the physiotherapist, as a member of the health care staff, should evaluate and analyze the amount of muscle present in a subject and its direct/indirect association with different diseases, a fact that has started to be considered under the light of other aspects by reason of the relationships it has with certain pathologies where the muscles are controllers of the homeostasis (4). By the seventh and eight decade, the maximum contractile force decreases between 20 % and 40 %, with multiple factors leading to produce sp, as it is the case of the loss of skeletal muscle fibers secondary to the reduction of active motoneurons, low levels of physical activity, hormone changes, decrease of the total intake of calories and proteins, increase of the inflammatory mediators and factors that lead to an altered synthesis of proteins, among others. SP prevalence can extend from 30 % for > 60 years old and can increase as the percentage grows (5). There is also a relation between the muscle changes that negatively affect in the production of impact strength and the secretion of myokines in the bone (6). Changes in the bone include the decrease of the cellularity of the periosteum, loss of the osteocytes of the bone matrix, bone marrow fat infiltration, and periosteal response dimmed to the growing factors and to the parathyroid hormone (6, 7).
Therefore, the aim of this article is to review the present literature on the molecular processes related to the loss of muscle mass induced by aging, as well as its impact on the human body movement (hbm). From this approach, a contribution to appropriating knowledge that promotes effective interventions through exercise is aimed at, thus promoting the increase of knowledge on physiotherapy.
Materials and Methods
A review of the literature on the molecular causes of the loss of muscle mass induced by aging, as well as on its main implications on the hbm was conducted. Scientific articles in English and Spanish on the matter published between 2006 and 2016 in the databases Pubmed, Scielo and PEDro were selected and reviewed. The search was carried out combining with the boolean and operator the terms ‘muscle aging’, ‘exercise’ and ‘physical therapy’.
The search strategy was focused in key elements like population (older adults), study design (controlled randomized, quasi-experimental, cross-sectional, cohort studies), observed outcomes (presence of muscle atrophy, sarcopenia), and the conducted intervention (exercise and functionality training). Publications with no description of the state of health or of the intervention, with no specific clarity of the outcomes on population, and interventions targeted at a population with a certain concomitant pathology or publications on young adults, adolescent and children were excluded from the analysis.
The analysis was complemented with other electronic sources and books related to the issue to be developed. Information was systematized, thus allowing three parts: 1) aging and sarcopenia, 2) molecular pathways of the loss of muscle mass induced by aging, and 3) aging, exercise and functionality.
Results
Aging and Sarcopenia
Population aging is one of the major achievements of mankind, but it is also highlighted as one of the biggest challenges to confront. In this sense, the modern gerontologic studies started in North America in 1941, where Nathan Sock was pioneer by conducting the first researches on aging. Gerontology studies aging in all aspects, from the molecular biology to the socioeconomic analysis or the consequences of retirement; of which geriatrics derives, being this a young science oriented to the preventive, therapeutic and social aspects of the elder in health or in disease situation (8).
By understanding these two postulates, it can be also understood that old age is not a disease but a natural biological process that can make the subject more fragile and susceptible to suffering diverse health complications. Physiologic aging implies morphological changes (it affects cells, organs and tissues), changes in the functioning of all body systems, loss of the ability to adapt to stress situations and to recover. Therefore, it is considered a universal, unavoidable and irreversible process in which affectation in the body systems (i. e., the muscular system), in the body composition and in the functioning of organs is produced.
From this perspective, the detriment of the skeletal muscle function associated with age is defined as dynapenia (dyna refers to “power”, “force”, and penia refers to “poverty”, and is characterized by muscle weakness subsequent to SP (9,10,11). Functional compromise is expressed in quantitative terms in changes in the metabolic index, increase in the insulin resistance, drop of the glucose tolerance, loss of bone mass, decrease of the cardiocirculatory response, and increase of the fat mass, among others. From the physiological standpoint, SP can be produced by the decrease of contractile muscle elements, by the reduction of the total number of myofibriles (10 % at the age of 50 years old and above), decrease of the size of the type II muscle fibers, as well as by a loss of motor units (12).
In relation to the myofibrillar reduction, it starts about the age of 25 years old and by 80 years old, at an older age, at an annual average between 8 and 10 %. Until the age of 30 years old, strength is preserved, between the ages of 30 and 50 years old it starts to decrease about 15 %, and after this age, it loses each decade in 30 %. In addition to this, the fractional synthesis speed (fss) of the molecular proteins (sarcoplasmic structures, the heavy myosin chain (hmc), and actin) decreases gradually in elder people. The fss of the hmc is lower in the elderly. The fss of the hmc s lower in older adults, while the fss of the sarcoplasmic proteins does not deteriorate; besides, with age there is loss in the muscle strength and it correlates with a drop in the fss of the molecular proteins (4).
Moreover, there are determining factors such as changes in the central nervous system (cns), dysfunction of the peripheral nerve, alterations in the structure and function of the neuromuscular junction, fat infiltration, cellular and molecular modifications of the muscle fibers that limit the production of muscle strength and power. There is also a reduction of the satellite cells (responsible of regeneration), loss of excitability (decrease of the receptors), decline of the myosin protein content (reduction of the number of cross-bondings and of the maximum strength previous to the failure), rigidity (elasticity reduction), physical inactivity (affecting type I fibers), loss of mitochondrial content and function, and comorbidities such as cancer, diabetes and type II chronic heart failure (13, 14).
that It has been also established that lipofuscin (pigment associated with age) and fat settle in the muscle and the loss muscle tissue is then replaced by hard fibrous tissue, which is more noticeable in the hands, that look skinny and bony. Changes in the muscle tissue, combined with the changes in the cns make that the muscles have less tone and less ability to contract (15). Although the adult skeletal muscle is composed of totally differentiated fibers, it preserves the ability to regenerate as a response to an injury and to modify its contractile and metabolic properties; thanks to satellite cells residing in between the basal lamina and the plasmalemma, that are swiftly activated as a response to the appropriate stimuli. However, under pathologic conditions or during the aging process, regeneration can be prevented by the formation of fibrotic tissue that result in the functional muscle damage (16, 17).
Additionally, there are hormonal, metabolic, nutritional, immunological and molecular factors contributing to sp. Serum levels of testosterone and adrenal androgens decline with aging. These hormones inhibit the production of interleukins 1 and 6, causing an indirect catabolic muscle effect. Menopause is associated with the reduction of the circulating levels of 17-ß-estradiol, additionally, the decrease of the muscle performance during menopause appears with the reduction of the production of ovarian hormones. On the other hand, the levels of the growth hormone (gh) and of the insulin growth factor I (igf-i) decline with age. The secretion of the gh from the pituitary stimulates the peripheral secretion of “systemic” igf-i, thus stimulating the growth of new muscle tissue, however, this production is lower in obese individuals, indicating that fat might be a confounding factor in the relation between the reduction of the gh levels and sarcopenia (18).
Altogether, according to the 2010 report of the European Working Group on Sarcopenia in Older People, criteria for the diagnosis of sp are based on identifying the decrease of muscle mass, of muscle strength and endurance. In order to confirm the diagnosis, two of the three characteristics must be met; however, it should be determined if the presence of sp in the subject is due to age as primary cause, or secondary to inactivity (bed rest, a sedentary lifestyle, lack of fitness, or conditions of zero gravity), secondary to a disease (inflammatory disease, malignancy, endocrine disease, or associated to advanced organic failure in kidneys, heart, lungs and/or brain), or secondary to nutrition (inadequate diet intake of energy and/or protein, malabsorption, gastrointestinal disorders or the use of medication that causes anorexia) (19). Thus the importance of knowing the molecular pathways that cause SP as a marker of the body’s aging process.
Molecular Pathways of the Loss of Muscle Mass Induced by Aging
There are various molecular pathways related to the loss of molecular mass induced by aging. One of the most known is related to the cell damage implied in the production of free radicals (fr) or of reactive oxygen species (ros), very much associated to the oxidative stress; for this reason it is one of the causes of the progressive metabolic deterioration. fr are highly reactive with an unpaired electron and a short half-life, whose accumulation causes damage to the structural lipids, proteins and dna.
Oxygen (O2) is useful for life in most living beings (acts in the mitochondrial respiration), it can accept four electrons, giving rise to a water molecule, however, when reduction is partial, derivative reactive species are generated, thus producing the radical superoxide (O2-) which can give rise to hydrogen peroxide (H2O2) and to the radical hydroxyl (ho), the most toxic of all. The term fr refers to any molecule or atom containing at least one unpaired electron, very reactive to other molecules, conferring instability and a very short half-life. In this line, the radical superoxide or the radical hydroxyl are free radicals, while hydrogen peroxide is not. Therefore, in a more correct way, they are called reactive oxygen species (ros); although there are also species reactive to nitrogen (rns) derivative of the nitric oxide. Since there are no reparation systems, these oxidized proteins must be degraded. When the degradation systems, such as the proteasome, are not able to remove properly, either because of their excessive number or because of the deterioration of the proteasome, the oxidized proteins build up inside the cell, producing toxicity or cell death (20).
For its part, the potential consequences of the damaged induced by ros include alterations in the fundamental processes such as adn replication, disruption of the protein secondary and tertiary structures, and alterations in the integrity and permeability of the membranes. The muscle production of fr accelerates with age due to a declination in the mitochondrial function and an increase in the electron displacement towards the transport chain, thus generating the mitochondrial production of ros. Since the mitochondria is the main source of ros, it is susceptible to oxidative damage and to the subsequent dysfunction, creating a vicious circle in which the ros mitochondrial production develops into oxidative damage, mitochondrial dysfunction (inefficient energy production and in the involvement of the muscle function), thus causing reduction in the muscle function (contractile dysfunction, regulatory changes, and muscle protein degradation) (19).
Other possible causes related to muscle aging are the mitochondrial dysfunction and the accumulation of damaged mitochondria (21). In animal models, mitofusin 2 (Mfn2) plays a key role in mitochondrial muscle damage control, since in the aging process there is a progressive Mfn2 reduction in the skeletal muscle of the mice, besides, the Mfn2 ablation produces a gene mark of aging. Additionally, the analysis with mice deficient in muscle Mfn2 showed that this condition lies behind alterations related to metabolic homeostasis and SP (2). Mfn2 deficiency reduced here autophagy and damaged the mitochondrial quality, thus leading to an exacerbated mitochondrial dysfunction. Curiously, Mfn2 deficiency induced by aging provokes an adaptative signal pathway dependent on ros trough the induction of HIF1α and BNIP3 transcription factor that compensates for the loss of mitochondrial autophagy and minimizes mitochondrial damage (2).
The lack of Mfn2 predisposes mice to suffer glucose intolerance and insulin resistance (previous stages to type II diabetes). Beside this metabolic role, it was proved that Mfn2 is involved in autophagy and in the quality control of mitochondria (22,23,24). The drop of Mfn2 during aging causes reduction of autophagy and mitophagy, thus contributing to a failure in the function of the mitochondrial recycling process and to a build-up of mitochondria in bad condition. These mitochondria present a deficit in their breathing capacity, a lower oxidative metabolism and increase of the oxidative stress, all associated to the build-up of damaged mitochondria that serve as a signal for the stabilization of the HIF1α transcription factor that activates the expression of the bnip3 protein. This protein is involved in the mitophagy or specific mitochondria degradation through autophagy (25).
According to Bujak, in 2015, the protein kinase activated by amp (ampk) accelerates autophagy, but its role in aging and in the muscle function induced by fasting is not known in great depth. Fasting mice that have no amp (ampk-mko) skeletal muscle develop hypoglycemia and hyperketosis. This is not due to the defective oxidation of the fatty oils, but to a blockage of the proteolysis that leads to a reduction of the circulating levels of alanine (an amino acid essential to gluconeogenesis). The markers of the muscle autophagy, including the Ulk1, Ser555 and Ser757 phosphorylation and the rfp-L3 aggregation are deteriorated. In consequence, with the autophagy alteration, the old ampk-mko mice present a significant myopathy, characterized by reduction of the muscle function, mitochondrial disease, and accumulation of protein autophagy. This corroborates the critical role for the autophagy played by the skeletal muscle ampk in the preservation of the blood glucose levels during a long-lasting fasting, as well as in maintaining the muscle integrity and the mitochondrial function during aging (26).
DeRuisseau in 2013 proved that the increase of the concentration of non-heme iron (nhi) observed in the skeletal muscle of old mice contributes to the development of SP. The highest nhi concentration in old age was consistent with the effects of the concentration of the muscle atrophy and the reduction of the heme iron levels, with no changes in the total iron content in the muscle (27). In 2015, Wang demonstrated the modifications in the phenotype of the myeloid cells that can influence the changes associated to old age in the muscle structure. They proved that muscle aging is associated to the increase of M2a macrophage anti-inflammatories that can increase muscle fibrosis (28). On his part, Du, in 2014, using analysis with micro-ray analysis, discovered that SP is associated to the increase of ARNm of the CCN1 cell matrix protein. ARNm of CCN1 regulates an increase of up to 113 times in old rats vs. young rats. When the muscle progenitor cells (mpc) were treated with recombinant CCN1, the muscle cell proliferation dropped. Curiously, it was found that the CCN1 made the p53, p16Ink4A and the pRP protein (hypophosphorilated retinoblastoma protein) levels grow, a fact that can stop cellular growth in mpc (29).
Both the skeletal muscle and the bone marrow contain myogenic stem cells (30,31,32,33,34). The population that lies in the muscles is heterogeneous, but their number dominate the typical satellite cells (mpc that migrate from the somites during the embryonic life). Another population is a group of multipotent mpc derivative of the bone marrow which are traced back by a gradient of growth factors released from the muscle during an injury or when exercising. Cells derivative from the bone marrow change their phenotype gradually, becoming muscle stem cells and can possibly reach the cell position and the Pax7 protein expression. The mesenchymal stem cells directly isolated from the bone marrow show also myogenic potential (35).
It has been proved that the satellite cells are heterogenous and that the subpopulations of these cells are able to carry out asymmetric divisions to produce myogenic progenitors or symmetric divisions to expand the collection of satellite cells. Therefore, a balance between extrinsic and intrinsic signals is necessary to control the progression and cell destiny determination cycle. Regulation shortcomings, as it can be observed in the degenerative conditions such as aging, can be harmful for the muscle regeneration (36, 37). Recent studies on biology of the skeletal muscle and the satellite cells in aging have established a critical impact of the systemic factors and of the functionality of the stem cells, whose capacity to regenerate and increase their regenerative potential when it is exposed to a favorable young environment (38,39,40) is detectable.
On the other hand, in 2012, Ramirez exposed that the tumoral necrosis factor α (tnf α) is a relevant mediator in the atrophy induced by aging. tnf α is a sarcoactive cytokine, associated to the inflammatory response and the muscle adaptation process. It is produced by monocytes, macrophages and B and T lymphocytes, and muscle cells. Studies in humans showed that the high tnf α concentration has a catabolic effect in systemic conditions, such as aging. In mice, the tnf α intramuscular injection produces degradation of myoproteins and inhibition of muscle differentiation, where the role of this cytokine suggests muscle atrophy. The catabolic effect of tnf α seems to be mediated by the nuclear factor kappa β36 and mitogen activated protein kinases pathways. The created proinflammatory stimulus produces the nuclear translocation of p65 (family member of the NFkB transcription factors), increases the expression of MuRF1, a protein involved in the balance of the titine and therefore in the organization of the sarcomere. Also, the increase of concentration of tnf α activates the p38, kinase protein activated by mitogens (p38mapk) that produces a rise in the expression of atrogin 1 (protein present in the muscle atrophy) (3, 41).
Recently, the genetic determination has allowed the association of the drop of muscle mass and the muscle strength in older people, these two risk phenotypes are widely recognized and studied in the sp, with a heritability ranging from 30 to 85 % for muscle strength, and from 45 to 90 % for muscle mass. It is well known that the development and maintenance of muscle mass in the first adult age reduces the risk of developing SP and leads to a healthy aging. For this reason, it seems to be important to identify which genetic factors interact with aging and specifically with the musculoskeletal response to exercise in those individuals (42,43,44).
Therefore, it is necessary to determine the molecular causes and consequences of sp. The present analysis and evaluation methods aim at determining with greater accuracy which variable (muscle mass, muscle strength or muscular endurance) does influence sp. In order to evaluate muscle mass, the use of anthropometry, computerized axial tomography, magnetic resonance, bioimpedance and dual x-ray absorptiometry is suggested. For measuring the muscle strength, manual dynamometry, measurements of total or partial body potassium vs. non-fatty soft tissue, knee flexion/extension, and maximum expiration flow are applied. To assess the muscle endurance, the short version of the questionnaire on physical performance, the stand up and walk test, the climbing stairs endurance test, and the programed raising and lowering test are used (19).
All these tests provide general and useful guidance for the physiotherapist when interpreting the involvement caused by sp, thus making it possible to implement the intervention strategy with the most suitable parameters according to the specific needs of the individual. The interventions based on the best available evidence of the appropriate prescription of the exercise, that is monitored and oriented by this professional, are derived from a deep analysis of the obtained results.
Aging, Exercise, and Functionality
For the physiotherapist, to understand and intervene the existent relation between aging and functionality is one of the main purposes that underlie his/her knowledge of the hbm. The project “El papel de la fisioterpia en la atención de las personas de la tercera edad” (The Role of the Physiotherapist Attending the Elderly) developed by the World Confederation for Physical Therapy together with the World Health Organization, recognizes the need of this professional to provide an effective physiotherapy service to the elderly based in knowledge and the techniques related to prevention and treatment of movement disorders that older people, with a clear understanding of the psychologic, social and environmental consequences of the aging process (45).
Primary aging is the progressive and unavoidable process characterized by the deterioration of the body and the skeletal muscle, it causes energetic mitochondrial deficiencies and reduces muscle mass. Secondary aging refers to structural and functional changes caused by age, diseases and lifestyle. Secondary aging can exacerbate the deficits in the mitochondrial function and the muscle mass, leading to insulin resistance. Practicing exercise counteracts the damaging effects of secondary aging, preventing the decrease of the mitochondrial breathing, mitigating muscle aging and increasing insulin sensitivity (46). The skeletal muscle is a dynamic tissue with a noticeable capacity to respond continuously to imposed stimuli (such as exercise) and, although scientific efforts have been made to better understand the underlying mechanism that controls the regeneration of the skeletal muscle, there has been poor impact on the clinical approaches used to treat muscle injuries and aging (47).
For the independence in developing ea it is indispensable to practice exercise to maintain adequate muscle mass and strength (48). The skeletal muscle undergoes important changes with age and SP has relevant metabolic repercussions that should be tempered (48 ,49). There is no clear cause for its onset, multiple mechanisms have been proposed to have relation with its development (50, 51). Older people have reduced capability to induce muscle hypertrophy with endurance exercise, however, it is very difficult to differentiate between what is physiological loss and what is atrophy due to inactivity (52, 53). A study conducted with Swedish athletes older than 65 years old showed that only those who performed strength training (weight lifting) were able to maintain the characteristics of the young muscle. However, athletes doing endurance training had the same muscle hypertrophy and a similar decline of the enzymatic capacity that sedentary individuals, proving that muscle strength reduction is conditioned by the functional limitations of the subject (4).
These changes have great repercussions at a functional level that can affect lq of the elder person. The diaphragm is the main inspiratory muscle, therefore, the knowledge of SP of it is critical for establishing the effects of aging in the respiratory tracks and the functional activities. Changes at the respiratory level have been found, as in 2013 Grising demonstrated using an animal model where the SP of the diaphragm shows a maximum specific strength of <34 % in old mice compared with young mice (54). Although initially the thinking was that the increase of the oxidative damage was the key of the ma, now it is believed that the ros could be one of the multiple components of the degenerative processes in this particular process. The skeletal muscle shows important rapid adaptations to ros produced by mitigated contractions in aged organisms; additionally, transgenic studies have indicated that overcoming these mitigated responses could prevent this loss (55). Reduction in the number of fibers can be caused by an irreversible damage, part of the fibrillar population seems to undergo a denervation process, although the re-innervation process is possible with aging (56).
The declining of strength is due in part to the reduction of the maximum voluntary activation of the agonists and to reduction in the coactivation of the antagonists, especially during dynamic actions. In the hbm the time needed to produce a certain submaximum strength level could be more important than the absolute strength level (18). Death associated to the age of the motor nerves and the subsequent loss of muscle fibers could explain the loss of strength associated to aging. The loss of muscle fibers is accompanied by the reduction of the randomness in distribution of the fiber type within the muscles, particularly of the type II denervated fibers that would be reinervated by the type I motor nerves. Due to the fact that weaker muscles with lower mass and functionally involved are more susceptible to muscle damage, reduction of the muscle strength associated to age is an important factor that affects the elder’s susceptibility to muscle damage induced by exercise. Since training strength enhances significatively the muscle mass in elder people, regular training could be an important preventive measure against muscle damage (18).
Some traditional proposals about exercise, such as swimming walking and cycling, do not reach the minimum threshold of physical demand able to stimulate protein biosynthesis. It is obvious to remind about being cautious when dosing physical workloads in older people since, at first, they will not be apt to tolerate certain efforts. Slow progression both in volume and in intensity should follow a process and planification, so to say, a meso-cycle of at least three months with efforts not higher than 30-40 % 1RM (adaptative phase), then moving (also being cautious according to possibilities) to more advanced, suggested performing levels (about 70 % 1RM). There is also a normal functioning threshold for different organs and systems, at 30 % of the maximum functional capacities. It is known that the frailty syndrome (common in old age) takes the form of a vicious circle constituted by SP, muscle weakness, gait disorders, physical inactivity, decline of the metabolic index and of the energy expenditure, and chronic malnutrition. According to statistics, between 10 and 25 % of the 65 years old and older individuals can be considered as fragile, reaching 46 % by 85 years old; this physiologic vulnerability results from a deterioration of the homeostatic reserves that will end in a lower response to stress (4).
SP is predictor of morbi-mortality in the elder and is associated to extremely high costs in the health care systems due to the reduction of the general physical function. Falls are the main cause of injury, morbidity and mortality in the elderly, commonly related to weakness of the toe, knee and hip muscles. Characteristically, a reduction of the maximum voluntary torque is produced in the higher and lower bodies, muscle power declines also with age, even to a higher extent than the peak torque, resulting in the deterioration of the physical function. Loss of strength in the quadriceps means that many older persons will have a strength level lower than the needed to get up of bed or to use their arms for help. The capacity to walk up or down stairs, to rise from a chair, or to use the bathtub could be seriously affected if the strength levels are not high enough, thus affecting lq (57).
Muscle mass becomes the biggest protein source of the body, representing 45 % of total body weight, therefore its valuable metabolic and functional contribution. The antigravity muscles as well as those of the lower limbs, loss their strength at twice the speed as the smaller muscles. It is in the legs where the most important muscle declines take place, they carry the loss of the functional, thermal-regulatory and immune capacities, as well as the deterioration of the nutritional status. With aging and very low levels of physical activity, muscle strength is a critical component of the ability to walk, here lies the importance of underlying strength training works in the lower body for the multiple motor, organic and physiological repercussions in this population. There are reports on an increase in the muscle protein synthesis (±50 %) accompanied by gains in strength after 16 weeks progressive strength training in men aged between 65 and 75 years old. Finally, physic inactivity is a significant factor that contributes to SP associated to aging. It is well established that those men and women who are physically less active have less muscle mass and an increased prevalence of inability (58).
Skeletal muscle health depends on the optimal function of its mitochondria. With advanced age, decreases in many mitochondrial variables are evident in the muscle, therefore the interest of developing interventions to reduce sp (59). An area that recently has attracted attention is the use of myogenic stem cells for bettering muscle regeneration, however, the fundamental role of the satellite cells in the homeostasis of normal aged muscles and the incidence in the endogenous regeneration of old muscles is still under research (60). Evidence is mounting of the benefits of exercise in people more susceptible to injuries, such as older adults. There seems to be consensus that exercise can be safe in aging, but exercise is generally recommended with an individualized prescription (frequency, intensity and duration) and type (endurance vs. aerobic), and requires attention by a physiotherapist since there is a vast diversity in the functional level and the comorbidities in elderly (61).
It is becoming clear that transference of strength from within and among the fibers plays also an important role in this process. This implies a secondary matrix of proteins that line up and transmit the strength produced by thick and thin filaments along the muscle fibers and toward the extracellular matrix; these specialized protein networks of the cytoskeleton help the strength that passes through the muscle and serve to protect the individual injury fibers (62). Old age individuals have an oxidative capacity near 50 % per muscle volume, over younger adults (63).
Another controversial question is whether aging affects the basal capillarization of the skeletal muscle and the angiogenesis; however, a study carried out by Gavin in 2015 confirmed that aging (age range between 57 and 76 years old) does not affect the muscle angiogenesis as a response to physical training in women who completed eight weeks of aerobic training by measuring muscle capitalization, interstitial vascular endothelial growth factor, and microvascular blood flow; although gender differences between men and women trained in similar ways could be found (64). Older adults that participate in 20-30 minutes exercise of moderate intensity in almost all days of the week have a better physical function than older people who are active throughout the day or who are inactive. Any kind of physical activity is better than none for protecting against functional limitations, but exercise confers a higher benefit for physical capacity (65).
Presently it is well known that physical exercise in older adults has proved benefits specially in increasing muscle mass and power, bettering functional parameters, such as capacity and walk speed, walking upstairs, and increasing the independence level in carrying out different activities of daily life (66, 67). Generally speaking, exercise and physical activity improve the muscle, osteoarticular, cardiocirculatory, pulmonary, endocrine-metabolic, immune and psychoneurological function, thus contributing to improve the functionality, a better adaptative response and a higher resistance to disease (67).
Conditioning of strength will lead to an increase of the muscle size due largely to the results of the increase of contractile proteins. It has been proved that these eccentric muscle contractions produce an ultrastructural injury (microscopic tears in the contractile muscle cells of the proteins) that can stimulate the increase of the renovation of the muscle protein. This muscle damage produces a cascade of metabolic events similar to an acute-stage response, and includes the activation and mobilization of the neutrophils, the increase of the interleukin-1 of the skeletal muscle, accumulation of macrophages, and increase in the synthesis and degradation of muscle proteins. Although resistance exercise increases the oxidation of essential amino acids and the need of proteins in the diet, resistance exercise provokes a decline of the nitrogen excretion, thus reducing the need of proteins in the diet. Research has shown that the increase of protein intake in the diet (up to 16 gr protein kg-1xday-1) can better the hypertrophic response to resistance exercise. It has also been proved that in elderly the use of protein supplements was associated to higher strength and muscle mass gains (68, 69).
While reduced mobility and functionality become more and more prevalent in the elderly, there is still a long way to go about sp treatment. Currently there are limited proposals that promote the elderly’s functionality through exercise, such as the geriatric park proposed by Martin Rodriguez, Lara Saez and Liria Lopez in 2007 (70). According to the report of the European Working Group on Sarcopenia in Older People, few clinical trials to test possible treatments are in progress. For this reason, they recommend three main variables of the results in the interventions: muscle mass, muscle strength and muscle resistance; the resting results can be considered secondary and of particular interest in specific research areas, such as everyday activities, lq, metabolic and biochemical markers; markers of inflammation, falls, admission to retirement home or to hospital, social assistance and/or mortality (19).
In relation to the exercise prescription, Frankel, Bean and Frontera recommend, for purposes of training physical qualities in older adults, to do exercise of moderate to low intensity between two to three times a week, it can be started with progressive resistance of low intensity in not trained or not active individuals (71). For resistance, it is recommended to carry out activities that corresponds to >5-6 met for 30 minutes five days a week, as well as maintaining a heart rate of between 60 and 70 % of the maximum projected or Borg scale, level of effort by 11-13; for balance, Tai Chi or exercises of proprioceptive training for toes three to four times a week combined with the other exercises. For flexibility it is suggested to carry out four to five repetitions during 30 seconds each exercise, adding static and dynamic flexibility exercises.
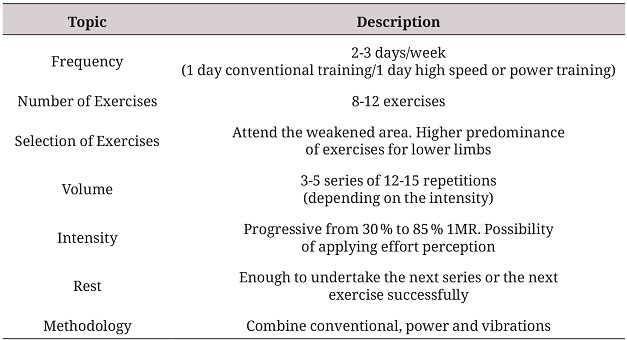
On their part, Mata Ordoñez and cols. specifically propose the following prescription of strength exercise in elderly (72).
Altogether, quoting the American School of Sports Medicine and the American Heart Association, Berdejo suggests to consider the following in the exercise programs for elderly:
To perform physical activity of aerobic nature of moderate intensity during at least 30 minutes, five days a week, or aerobic activities of vigorous intensity for 20 minutes, three days a week.
To combine moderate and vigorous intensities to achieve the minimum recommendations of physical activity.
At least two days in a week (not consecutive) to perform activities to develop muscle force and resistance. Development of big muscle groups and a moderate intensity are recommended, with a weight that allows 10 to 15 repetitions (8-12 repetitions) of 8 to 10 exercises. Breath should never be held (Valsalva maneuver) since it raises blood pressure.
For maintaining flexibility, to perform at least two days a week flexibility exercises during 10 minutes each day. Stretches should be maintained 10 to 30 seconds and repeated three to four times.
To perform exercises that maintain or better balance in order to reduce risk for falls.
When the older person is under medical treatment, the physical activities he/she performs should be safe for the corresponding treatment.
Physical activity performed by elderly should be planed and periodized. Additionally, they should be periodically evaluated in order to adapt planning or programming to their individual condition or physical level (73).
On this basis and according to the later scientific advances in the area of kinesiogenomics as a new research field in physiotherapy it has been shown that there are genes that promote the development of a high performance athlete that relate to resistance to physical effort; strength, mass and muscle power gaining; susceptibility to injuries and that are associated to specific psychologic responses. The found genes related to resistance are adrb2, 79C/G, bdkbr2, chrm2, EpoR, hbb, hif-1 α, gys1, nos3, nrf2, ppar5, vegf; the ones related to strength, muscle mass and power are ace, actn3, ampd1, ck-mm, igf 1. The genes associated to injury susceptibility are col1a1, col5a1, mmp3, tnc; and those connected to psychologic responses are 5htt, bdnf. Thanks to these discoveries the possibility to carry out genic therapies aiming at creating genetically gifted individuals can be proposed (74). Even if this has been discovered in high performance athletes, the continuous traversing of research could bring a promising future in the bettering of strength in the elderly as one of the possible beneficiaries of the improvement of the muscle strength.
Discussion
Nowadays we know more about the adaptability of diverse biologic systems, as well as the ways in which regular exercise influence them. Understanding and intervening the different muscle pathways that may lead to aging will allow that the physiotherapist develops effective and safe interventions for older adults, with the purpose of improving their lq and functionality in carrying out everyday life activities. Through an adequate exercise prescription substantial changes in the sp process could be done.
Training muscle strength help compensating the muscle mass and resistance which usually is associated to normal aging. Among the additional recognized benefits derived from regular exercising there are an improvement in the bone and articular conditions; a lower risk of suffering osteoporosis; an improvement in the postural stability and better balance and, therefore, a decline in the possibility of falling down and thus suffering from fractures; an increase in flexibility and in the range of motion; and the bettering and/or controls of certain metabolic profiles. Together to all these recognizable advantages it is good to add the implicit psychologic well-being that represents being able to dominate the own body at the more advanced ages, a fact that, when is impossible to concreate, incorporates a recurrent depression towards that incapacity for the elderly.
Including exercise in the program as an efficient way to prevent some diseases associated to aging is a challenge of the professional action that the physiotherapist must adopt. The training capacity in older individuals is evidenced in their capacity to adapt and respond to resistance and strengthening training. Aerobic efforts maintain and better aspects of cardiovascular functioning, such as VO2, cardiac output, and the arteriovenous O2 difference.
Exercise in older adults should be oriented towards an improvement of the muscle strength and the functional capacities, thus helping in certain diseases such as osteoporosis or hypertension. The moderate-high levels of exercise tend to improve the muscle strength, thus being a sufficiency indicator of the habitual levels in physical activity and its relation to the present health status. The challenge for physiotherapists is to identify the adequate exercise prescription for the strength training in elderly, thus adapting it to each individual situation according to the person’s capacities and needs.
Muscle regeneration provides a paradigm in physiotherapy to study how the extrinsic signals coordinate the cellular genic expression to maintain the muscle strength in the elderly. Extrinsic and/or intrinsic faults in cell regulation are considered determining for the decrease of the regenerative capacity and the precise understanding of the mechanisms controlling the satellite cell progression and the recruitment of other types of stem cells with myogenic potential it is crucial if it is aimed at exploiting the power of these cells in the fight against myopathic conditions caused by muscle aging.
It is important to continue to develop researches with high scientific level that allow understanding, evaluating, and intervening the mechanisms of the loss of muscle mass induced by aging in order to carry out individual interventions according to the particular needs, thus improving functionality and ql of the elderly, where these are one of the main targets of the physiotherapist’s action, thus contributing to the growth and strengthening of this profession. Understanding the molecular mechanisms involved in muscle aging is important for this professional because it allows comprehending the condition of the older adult in which he/she intervenes, understanding that physiotherapy uses intervention modalities that interact with the body tissues, on which a specific biological effect can take place.
Authors’ contributions
The author contributed with the revision, analysis and writing of the manuscript.
Acknowledgements
The author wants to thank the professors and leadership positions of the Manuela Beltran University in Bucaramanga for the support and motivation offered in the redaction process.
Responsibility Disclaimer
The author has no conflict of interests to declare.
References
1.Ramírez C. Importancia de la biología molecular para la fisioterapia moderna. Salud uis. 2011;43(3):317-20.
2.Sebastián D, Sorianello E, Segalés J, Irazoki A, Ruiz-Bonilla V, Sala D, et al. Mfn2 deficiency links age-related sarcopenia and impaired autophagy to activation of an adaptive mitophagy pathway. EMBO J. 2016;35(15):1677-93. Doi: 10.15252/embj.201593084
3.Ramírez C. Una visión desde la biología molecular a una deficiencia comúnmente encontrada en la práctica del fisioterapeuta: la atrofia muscular. Salud uis. 2012;44(3):31-39.
4.Roig JL. Sarcopenia: Algo más que la disminución de la masa muscular. PubliCE Standard [internet]. 2002 [citado 2017 oct 15] Disponible en: https://www.g-se.com/es/fisiologia-del-ejercicio/articulos/sarcopenia-algo-mas-que-la-disminucion-de-la-masa-muscular-231
5.Doherty TJ. Invited review: Aging and sarcopenia. J Appl Physiol (1985). 2003;95(4):1717-27. Doi: 10.1152/japplphysiol.00347.2003
6.Curtis E, Litwic A, Cooper C, Dennison E. Determinants of muscle and bone aging. J Cell Physiol. 2015;230(11):2618-25. Doi: 10.1002/jcp.25001
7.Fielding RA, Vellas B, Evans WJ, Bhasin S, Morley JE, Newman AB, et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc. 2011;12:249-56. Doi: 10.1016/j.jamda.2011.01.003
8.Rodríguez-Diaz O. Rehabilitación Funcional del anciano. MEDISAN [internet]. 2009 [citado 2018 sep 15];13(5). Disponible en: https://www.bvs.sld.cu/revistas/san/vol13_5_09/san14509.pdf
9.Sáez-Moreno MA, Jiménez Lorenzo R, Lueso Moreno M, García Atienza EM, Castaño M, López-Torres Hidalgo J. Dinapenia y función musculo-esquelética en los pacientes mayores de 65 años. Rev Clín Med Fam. 2018;11(1):8-14.
10.Cesari M, Fielding RA, Pahor M, Goodpaster B, Hellerstein M, Van Kan GA, et al. Biomarkers of sarcopenia in clinical trials-recommendations from the International Working Group on Sarcopenia. J Cachexia Sarcopenia Muscle. 2012;3:181-90. Doi: 10.1007/s13539-012-0078-2
11.Novotny SA, Warren GL, Hamrick MW. Aging and the muscle-bone relationship. Physiology. 2015;30:8-16. Doi: 10.1152/physiol.00033.2014
12.Manini TM, Hong SL, Clark BC. Aging and muscle: a neuron’s perspective. Curr Opin Clin Nutr Metab Care. 2013;16(1):1-10. Doi: 10.1097/MCO.0b013e32835b5880
13.Miljkovic N, Lim JY, Miljkovic I, Frontera WR. Aging of skeletal muscle fibers. Ann Rehabil Med. 2015;39(2):155-62. Doi: 10.5535/arm.2015.39.2.155
14.Plow EB, Cunningham DA, Bonnett C, Gohar D, Bayram M, Wyant A, et al. Neurophysiological correlates of aging-related muscle weakness. J Neurophysiol. 2013;110:2563-73. Doi: 10.1152/jn.00205.2013
15.Minaker KL. Common clinical sequelae of aging. Chapter 24 In: Goldman L, Schafer AI. Cecil Medicine. 24th Ed. Philadelphia: Saunders Elsevier; 2011.
16.Barberi L, Scicchitano BM, De Rossi M, Bigot A, Duguez S, Wielgosik A, et al. Age-dependent alteration in muscle regeneration: the critical role of tissue niche. Biogerontology. 2013;14:273-92. Doi: 10.1007/s10522-013-9429-4
17.Bentzinger CF, Wang YX, Dumont NA, Rudnicki MA. Cellular dynamics in the muscle satellite cell niche. EMBO reports. 2013;14:1062-72. Doi: 10.1038/embor.2013.182
18.Del Rosso S. Fisiología del envejecimiento. Módulo 1 del Posgrado de Actividad Física en Adultos Mayores [internet]. 2015 [citado 2017 oct 15]. Disponible en: https://www.g-se.com/es/actividad-fisica-y-entrenamiento-en-adultos-mayores/blog/envejecimiento-y-actividad-fisica
19.Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, et al. Sarcopenia: European consensus on definition and diagnosis Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010;39(4):412-23. Doi: 10.1093/ageing/afq034
20.Cabiscol E. Oxidación celular y envejecimiento. Radicales libres: doctor Jekyll y míster Hyde [on line]. 2014 [cited 2017 Oct 16]. Available From: https://www.sebbm.es/web/es/divulgacion/rincon-profesor-ciencias/articulos-divulgacion-cientifica/343-oxidacion-celular-y-envejecimiento-radicales-libres-doctor-jekyll-y-mister-hyde
21.Johnson ML, Robinson MM, Nair KS. Skeletal muscle aging and the mitochondria. Trends Endocrinol Metab. 2013;24(5):247-56. Doi: 10.1016/j.tem.2012.12.003
22.Zhao T, Huang X, Han L, Wang X, Cheng H, Zhao Y, et al. Central role of mitofusin 2 in autophagosome-lysosome fusion in cardiomyocytes. J Biol Chem. 2012;287(28):23615-25. Doi: 10.1074/jbc.M112.379164
23.Muñoz JP, Ivanova S, Sánchez-Wandelmer J, Martínez-Cristóbal P, Noguera E, Sancho A, et al. Mfn2 modulates the upr and mitochondrial function via repression of PERK. EMBO J. 2013;32(17):2348-61. Doi: 10.1038/emboj.2013.168
24.Chen Y, Dorn GW. PINK1-phosphorylated mitofusin 2 is a Parkin receptor for culling damaged mitochondria. Science. 2013;340(6131): 471-5. Doi: 10.1126/science.1231031
25.Sebastián D, Hernández-Álvarez MI, Segalés J, Sorianello E, Muñoz JP, Sala D, et al. Mitofusin 2 (Mfn2) links mitochondrial and endoplasmic reticulum function with insulin signaling and is essential for normal glucose homeostasis. Proc Natl Acad Sci USA. 2012;109(14):5523-8. Doi: 10.1073/pnas.1108220109
26.Bujak AL, Crane JD, Lally JS, Ford RJ, Kang SJ, Rebalka IA, et al. AMPK activation of muscle autophagy prevents fasting-induced hypoglycemia and myopathy during aging. Cell Metab. 2015;21(6):883-90. Doi: 10.1016/j.cmet.2015.05.016
27.DeRuisseau KC, Park YM, DeRuisseau LR, Cowley PM, Fazen CH, Doyle RP. Aging-related changes in the iron status of skeletal muscle. Exp Gerontol. 2013;48(11):1-19. Doi: 10.1016/j.exger.2013.08.011
28.Wang Y, Wehling-Henricks M, Samengo G, Tidball JG. Increases of M2a macrophages and fibrosis in aging muscle are influenced by bone marrow aging and negatively regulated by muscle-derived nitric oxide. Aging Cell. 2015;14:678-88. Doi: 10.1111/acel.12350
29.Du J, Klein JD, Hassounah F, Zhang J, Zhang C, Wang XH. Aging increases CCN1 expression leading to muscle senescence. Am J Physiol Cell Physiol 2014;306:C28-36. Doi: 10.1152/ajpcell.00066.2013
30.Kang JS, Krauss RS. Muscle stem cells in developmental and regenerative myogenesis. Curr Opin Clin Nutr Metab Care. 2010;13(3):243-8. Doi: 10.1097/MCO.0b013e328336ea98
31.Relaix F, Zammit PS. Satellite cells are essential for skeletal muscle regeneration: the cell on the edge returns centre stage. Development. 2012;139:2845-56. Doi: 10.1242/dev.069088
32.Cousin W, Ho ML, Desai R, Tham A, Chen RY, Kung S, et al. Regenerative capacity of old muscle stem cells declines without significant accumulation of dna Damage. PLoS ONE. 2013;8(5):e63528-38. Doi: 10.1371/journal.pone.0063528
33.Montarras D, L’honoré A, Buckingham M. Lying low but ready for action: the quiescent muscle satellite cell. FEBS Journal. 2013;280(2013):4036-50. Doi: 10.1111/febs.12372
34.Verdijk LB, Snijders T, Drost M, Delhaas T, Kadi F, Van Loon LJC. Satellite cells in human skeletal muscle; from birth to old age. Age. 2014;36:545-57. Doi: 10.1007/s11357-013-9583-2
35.Burdzińska A, Gala K, Pączek L. Myogenic stem cells. Folia Histochem Cytobiol. 2008;46(4):401-12. Doi: 10.2478/v10042-008-0060-1
36.Dumont NA, Wang YX, Rudnicki MA. Intrinsic and extrinsic mechanisms regulating satellite cell function. Development. 2015;142:1572-81. Doi: 10.1242/dev.114223
37.Blau HM, Cosgrove BD, Ho ATV. The central role of muscle stem cells in regenerative failure with aging. Nat Med. 2015;21(8):854-62. Doi: 10.1038/nm.3918
38.García-Prat L, Sousa-Victor P, Muñoz-Cánoves P. Functional dysregulation of stem cells during aging: a focus on skeletal muscle stem cells. FEBS Journal. 2013;280(2013):4051-62. Doi: 10.1111/febs.12221
39.Sousa-Victor P, Perdiguero E, Muñoz-Cánoves P. Geroconversion of aged muscle stem cells under regenerative pressure. Cell Cycle. 2014;13:20:3183-90. Doi: 10.4161/15384101.2014.965072
40.Jang YC, Sinha M, Cerletti M, Dall’Osso C, Wagers AJ. Skeletal muscle stem cells: Effects of aging and metabolism on muscle regenerative function. Cold Spring Harb Symp Quant Biol. 2011;76:101-11. Doi: 10.1101/sqb.2011.76.010652
41.Pereira AF, Silva AJ, Matos Costa A, Monteiro AM, Bastos EM, Cardoso Marques M. Muscle tissue changes with aging. Acta Med Port 2013;26(1):51-5. PMID: 23697358.
42.Jackson MJ. Redox regulation of muscle adaptations to contractile activity and aging. J Appl Physiol. 2015;119:163-71. Doi: 10.1152/japplphysiol.00760.2014
43.Karalaki M, Fili S, Philippou A, Koutsilieris M. Muscle regeneration: Cellular and molecular events. In Vivo. 2009; 23:779-96. Doi: 0258-851X
44.Deer RR, Volpi E. Protein intake and muscle function in older adults. Curr Opin Clin Nutr Metab Care. 2015;18(3):248-53. Doi: 10.1097/MCO.0000000000000162
45.Podestá O, Risso F. El Rol de los fisioterapeutas en la Tercera Edad. [Tesis de Grado] [Argentina]: Universidad Abierta Interamericana [internet]. 2009 [citado 2018 sept 15] Disponible en: https://www.imgbiblio.vaneduc.edu.ar/fulltext/files/TC098739.pdf
46.Cartee GD, Hepple RT, Bamman MM, Zierath JR. Exercise promotes healthy aging of skeletal muscle. Cell Metab. 2016;23(6):1034-47. Doi: 10.1016/j.cmet.2016.05.007
47.Ambrosio F, Kadi F, Lexell J, Fitzgerald GK, Boninger ML, Huard J. The effect of muscle loading on skeletal muscle regenerative potential: An update of current research findings relating to aging and neuromuscular pathology. Am J Phys Med Rehabil. 2009;88(2):145-55. Doi: 10.1097/PHM.0b013e3181951fc5
48.Crescenzo R, Bianco F, Mazzoli A, Giacco A, Liverini G, Iossa S. Skeletal muscle mitochondrial energetic efficiency and aging. Int. J. Mol. Sci. 2015;16:10674-85. Doi: 10.3390/ijms160510674
49.Rivas DA, Lessard SJ, Rice NP, Lustgarten MS, So K, Goodyear LJ, et al. Diminished skeletal muscle microrna expression with aging is associated with attenuated muscle plasticity and inhibition of IGF-1 signaling. FASEB J. 2014;28:4133-47. Doi: 10.1096/fj.14-254490
50.Salech F, Jara R, Michea L. Cambios fisiológicos asociados al envejecimiento. Rev. Med. Clin. Condes. 2012;23(1):19-29. Doi: 10.1016/S0716-8640(12)70269-9
51.Olmos Martínez JM, Martínez García J, González Macías J. Envejecimiento músculo-esquelético. REEMO. 2007;16(1):1-7. Doi: 10.1016/S1132-8460(07)73495-5
52.Kennis E, Verschueren S, Van Roie E, Thomis M, Lefevre J, Delecluse C. Longitudinal impact of aging on muscle quality inmiddle-aged men. Age. 2014;36:9689-701. Doi: 10.1007/s11357-014-9689-1
53.Demontis F, Piccirillo R, Goldberg AL, Perrimon N. Mechanisms of skeletal muscle aging: insights from drosophila and mammalian models. Disease Models & Mechanisms. 2013;6:1339-52. Doi: 10.1242/dmm.012559
54.Greising SM, Mantilla CB, Gorman BA, Ermilov LG, Sieck GC. Diaphragm muscle sarcopenia in aging mice. Exp Gerontol. 2013;48(9):881-7. Doi: 10.1016/j.exger.2013.06.001
55.Jackson MJ. Interactions between reactive oxygen species generated by contractile activity and aging in skeletal muscle? Antioxid. Redox Signal. 2013;19(8):804-12. Doi: 10.1089/ars.2013.5383
56.Häkkinen K, Alen M, Kraemer WJ, Gorostiaga E, Izquierdo M, Rusko H, et al. Neuromuscular adaptations during concurrent strength and endurance training versus strength training. Eur J Appl Physiol. 2003;89(1):42-52. Doi: 10.1007/s00421-002-0751-9
57.American College of Sport Medicine. ACSM’s Guidelines for Exercise Testing and Prescription. 9th Edition. Philadelphia: Lippincott Williams & Wilkins; 2013.
58.Guasconi V, Puri PL. Chromatin: the interface between extrinsic cues and the epigenetic regulation of muscle regeneration. Trends Cell Biol. 2009;19(6):286-94. Doi: 10.1016/j.tcb.2009.03.002
59.Grounds MD. Therapies for sarcopenia and regeneration of old skeletal muscles. BioArchitecture. 2014;4(3):81-7. Doi: 10.4161/bioa.29668
60.Carter HC, Chen CCW, Hood DA. Mitochondria, Muscle Health, and Exercise with advancing age. Physiology. 2015;30:208-23. Doi: 10.1152/physiol.00039.2014
61.Lovering RM, Brooks SV. Eccentric exercise in aging and diseased skeletal muscle: good or bad? J Appl Physiol. 2014;116:1439-45. Doi: 10.1152/japplphysiol.00174.2013
62.Hughes DC, Wallace MA, Baar K. Effects of aging, exercise, and disease on force transfer in skeletal muscle. Am J Physiol Endocrinol Metab. 2015;309:E1-E10. Doi: 10.1152/ajpendo.00095.2015
63.Evans WJ. Exercise strategies should be designed to increase muscle power. J Gerontol A Biol Sci Med Sci. 2000;55(6):309-10. Doi: 10.1152/ajpendo.00095.2015
64.Gavin TP, Kraus RM, Carrithers JA, Garry JP, Hickner RC. Aging and the skeletal muscle angiogenic response to exercise in women. J Gerontol A Biol Sci Med Sci. 2015;70(10):1189-97. Doi: 10.1093/gerona/glu138
65.Brach JS, Simonsick EM, Kritchevsky S, Yaffe K, Newman AB. The association between physical function and lifestyle activity and exercise in the health, aging and body composition study. J Am Geriatr Soc. 2004;52(4):502-9. Doi: 10.1111/j.1532-5415.2004.52154.x
66.Jara R. Efectos del ejercicio en adultos mayores. Rev Hosp Clín Univ Chile. 2015;26:293-9.
67.Landínez-Parra NS, Contreras-Valencia K, Castro-Villamil A. Proceso de envejecimiento, ejercicio y fisioterapia. Revista Cubana de Salud Pública 2012;38(4):562-80. Doi: 10.1590/S0864-34662012000400008
68.Evans WJ. Effects of exercise on senescent muscle. Clin Orthop Relat Res. 2002;403:S211-20. Doi: 10.1097/00003086-200210001-00025
69.Conley KE, Jubrias SA, Esselman PC. 2000. Oxidative capacity and ageing in human muscle. J Physiol 2000;526(1):203-10. Doi: 10.1111/j.1469-7793.2000.t01-1-00203.x
70.Rodríguez-Martín CR, Sáez-Lara CB, López-Liria R. El parque geriátrico: fisioterapia para nuestros mayores. GEROKOMOS 2007;18(2):84-8.
71.Berdejo D. Prescription of physical activity for old people in relation to their various diseases. J Sport Health Res. 2009;1(2):101-11.
72.Mata-Ordóñez F, Chulvi-Medrano I, Heredia-Elvar JR, Moral-González S, Marcos-Becerro JF, Da Silva-Grigogolleto ME. Entrenamiento de la fuerza y sarcopenia. Evidencias actuales. Journal of Sport and Health Research 2013; 5(1):7-24.
73.Frankel JE, Bean JF, Frontera WR. Exercise in the Elderly: Research and clinical practice. Clin Geriatr Med. 2006; 22(2006): 239-256. Doi: 10.1016/j.cger.2005.12.002
74.Alfonso-Mantilla J, Martínez-Santa J, Vargas O. Kinesiogenómica: Una nueva perspectiva de investigación en fisioterapia. Rev Mov Cient. 2016;10(1): 78-86. Doi: 2011-7191
Author notes
* Corresponding author: ivandpr@hotmail.com