
ISSN:1692-7273 | eISSN:2145-4507



Environmental Risk Factors and Acute Lymphoblastic Leukaemia in Childhood
Factores de riesgo ambientales y leucemia linfoblástica aguda en la infancia
Fatores de risco ambientais e leucemia linfoblástica aguda na infancia
Fernanda Alves Cangerana Pereira, Antônio Pedro Mirra, Maria do Rosário Dias de Oliveira Latorre, João Vicente de Assunção
Environmental Risk Factors and Acute Lymphoblastic Leukaemia in Childhood
Revista Ciencias de la Salud, vol. 15, no. 1, 2017
Universidad del Rosario
Fernanda Alves Cangerana Pereira 1 facan@usp.br
University of São Paulo, Brasil
Antônio Pedro Mirra 2
University of São Paulo, Brasil
Maria do Rosário Dias de Oliveira Latorre 3
University of São Paulo, Brasil
João Vicente de Assunção 4
University of São Paulo, Brasil
Received: 07 January 2016
Accepted: 24 October 2016
Additional information
To cite this
article: Cangerana FA, Mirra AP, Dias de Oliveira MR, de Assunção JV.
Environmental risk factors and acute lymphoblastic leukaemia in childhood. Rev
Cienc Salud. 2017;15(1):129-144. Doi: https://doi.org/10.12804/revistas.urosario.edu.co/revsalud/a.5386
Abstract: Objectives: To verify the paper of environmental risks in the process of developing Acute lymphoblastic leukemia (ALL) in childhood. Materials and methods: A search was conducted on scientific literature in order to evaluate the role of environmental risk factors in acute lymphoblastic leukemia development. Results: The environmental role in development of ALL in children is unknown. The two steps theory proposed by Greaves in 1988 is the most acceptable hypothesis. One first event occurs in uterus or near the moment of birth; this mutation induces the development of pre-leukemia clones. Another event, happening in the early infancy, would develop this first pre-leukemia clone into clinical leukemia. Based on the environmental events linked to other types of leukemia and in other studies, some risk factors have been proposed as inducing the mutations leading to ALL. Radiation (ionizing or not), infectious agents, and chemical products are thought to be the responsible for the disease. In the last decades, many authors tried to found an association between these risk factors and the development of childhood ALL. The results however were inconclusive and even contradictories. Ionizing radiation is the only one proved to play a role in the development of leukemia. Conclusion: As leukemia is a disease that affects children, costs of this pathology are high when talking about money expend for diagnosis and treatment and when thinking on the social and psychological burden. Considering the theory proposed by Greaves, there are at least, two moments, when the patient should avoid the exposition that leads to leukemia. Therefore, it is very important to find the real risk factor or factors that are involved in the process.
Keywords: Leukemia, environmental factors, childhood.
Resumen: Objetivos: Verificar el papel de los riesgos ambientales en el proceso de desarrollo de la leucemia linfoblástica aguda (LLA) en la infancia. Materiales y métodos: Se realizó una búsqueda en la literatura científica con el fin de evaluar el papel de los factores de riesgo ambientales en el desarrollo de la leucemia linfoblástica aguda. Resultados: El papel del medio ambiente en el desarrollo de la LLA en niños es desconocida. La teoría de dos etapas propuesta por Greaves, en 1988 es la hipótesis más aceptable. Un primer evento se produce en el útero o cerca del momento del nacimiento; esta mutación induce el desarrollo de clones pre-leucemia. Otro acontecimiento, suceso en la infancia temprana, se desarrollaría este primer clon de pre-leucemia en leucemia clínica. Con base en los eventos ambientales relacionados con otros tipos de leucemia y en otros estudios, algunos factores de riesgo se han propuesto como la inducción de las mutaciones que conducen a LLA. Radiación (ionizante o no), agentes infecciosos, y productos químicos se cree que son los responsables de la enfermedad. En las últimas décadas, muchos autores intentaron encontrado una asociación entre estos factores de riesgo y el desarrollo de la LLA infantil. Sin embargo los resultados no fueron concluyentes e incluso contradictorias. La radiación ionizante es el único demostrado desempeñar un papel en el desarrollo de la leucemia. Conclusión: Como la leucemia es una enfermedad que afecta a los niños, los costos de esta patología son altos cuando se habla de dinero gastar para el diagnóstico y tratamiento y cuando se piensa en la carga social y psicológico. Teniendo en cuenta la teoría propuesta por Greaves, hay por lo menos dos momentos cuando el paciente debe evitar la exposición que conduce a la leucemia. Por lo tanto, es muy importante encontrar el factor de riesgo real, o factores, que están involucrados en el proceso.
Palabras clave: leucemia, factores ambientales, infancia.
Resumo: Objetivos: Para verificar o papel dos riscos ambientais no processo de desenvolvimento da leucemia linfoblástica aguda (LLA) na infância. Materiais e métodos: realizou-se uma busca na literatura científica com o fim de avaliar o papel dos fatores de risco ambientais no desenvolvimento da leucemia linfoblástica aguda. Resultados: O papel do meio ambiente no desenvolvimento da LLA em crianças é desconhecido. A teoria de duas etapas proposto por Greaves, em 1988 é a hipótese mais aceitável. Um primeiro evento se produz “no útero” ou perto do momento do nascimento; esta mutação induz o desenvolvimento de clones pré-leucemia. Outro acontecimento, sucedido na infância precoce, se desenvolveria este primeiro clone de pré-leucemia em leucemia clínica. Com base nos eventos ambientais relacionados com outros tipos de leucemia e em outros estudos, alguns fatores de risco se têm proposto como a indução das mutações que conduzem a LLA infantil. Radiação (ionizante ou não), agentes infecciosos, e produtos químicos acredita-se que são responsáveis da doença. Nas últimas décadas, muitos autores tentaram encontrar uma associação entre estes fatores de risco e o desenvolvimento da LLA infantil. No entanto, os resultados não foram concludentes e inclusive contraditórios. A radiação ionizante é o único demonstrado desempenhar um papel no desenvolvimento da leucemia. Conclusão: Como a leucemia é uma doença que afeta às crianças, os custos desta patologia são altos quando se fala de dinheiro gasto para o diagnóstico e tratamento, e quando se pensa na carga social e psicológica. Tendo em conta a teoria proposta por Greaves, há pelo menos dois momentos quando o paciente deve evitar a exposição que conduz à leucemia. Pelo tanto, é muito importante encontrar o fator de risco real, ou fatores, que estão envolvidos no processo.
Palavras-chave: Leucemia, fatores ambientais, infância.
Introduction
Leukemia is a group of hematological diseases arising from the transformation of a blood pluripotent stem cell, or a mature cell, in some cases, in the bone marrow. This cell grows more than the normal cells in marrow, liver, spleen, and lymph nodes. Hematological neoplasms include heterogeneous diseases with different cytology, histology, epidemiology, etiology, and prognoses. In several countries, leukemia represents 30% of neoplasms occurring in childhood. In São Paulo (Brazil), according to the Sao Paulo Cancer Register, leukemia represents 20.2% and 31.3% of neoplasms in females and males respectively, and the most frequent leukemia is acute lymphoblastic leukemia (ALL). Between 1997 and 2008, the incidence of this disease was 5.1, 3.9, and 3.6 per 100.000 females (for 0-4 years old, 5-9, and 10-14 years old, respectively) and 6.8, 5.0, and 4.3 for males in the same age cohorts (1, 2).
The precursor to this disease can be a T or a B lymphocyte. B cell precursor leukemia represents 80-85% of all cases of all. It is most frequent in children under six years of age. T-precursor leukemia makes up about 15% of the cases and is more common in teenagers than in young children. In the case of B-precursor leukemia, the cure rate rises to 80% of cases. However, there are still some types of the disease with poor prognoses and low rates of remission. Increased risk groups are recognized by the cytogenetics, genotype, and immunophenotype of the malignant cell (1).
In the United States, all distribution peaks between 2-5 years of age at 7.5-8.5 cases per 100,000. The incidence declines to 1.5-2.0 cases per 100,000 at about 10 years of age. This peak was detected in industrialized countries in the first years of the twentieth century and has not been detected yet in non-industrialized countries. The incidence of childhood cancer has increased in economically developed countries in recent decades, and the increase is unlikely to be an artefact. Causes of the increase have not been identified, but preventable environmental exposure may play a role (3, 4, 5).
More than 200 genetic alterations have been linked to leukemia, including point mutations, deletions, and inversions. Ikaros is a gene whose activity is essential for normal hematopoiesis. It acts as a master regulator of lymphoid and myeloid development as well as a tumor suppressor. It is crucial for controlling the development and functioning of almost all hematopoietic cell types and has been found to activate or repress transcription, depending on the target gene. Mutations or polymorphisms that lead to reduced Ikaros function or expression have also been found to be a major genetic feature in human B-cell acute lymphoblastic leukemia (B-ALL) (6, 7, 8).
Several studies have shown that genetic alterations in leukemia occur in utero. Many of these genetic alterations occur in healthy populations. Screening of newborn cord blood samples suggests that around 1% have the TEL-AMI1 fusion gene. The frequency of these alterations is much higher than the risk of developing leukemia. It shows that this first event does not induce leukemia alone and that ALL development is a process entailing at least two events. This finding accords with the theory proposed by Greaves in 1988. While the first event occurs in utero and is more common, the second event is probably rare and determines the incidence of the disease. Greaves suggests that for each child that develops leukemia, there are 20 healthy children who had the first mutation in utero, had the fusion gene, and generated pre-leukemic clones in marrow (6, 9, 10, 11, 12).
Methods
A literature review was conducted to determine the environmental risk factors linked to all and determine the state of art of this subject. PubMed and LILACS were consulted to find papers that evaluated environmental exposure and acute lymphoblastic leukemia. Inclusion criteria were adopted to include studies in which patients were between zero and fifteen years of age. Every year and country of publication was included, since these factors do not affect the evaluation of the causal relationship. We selected papers that dealt with radiation, pesticides, and the infectious theory to explain the development of ALL, because these factors were the most frequently found in our search. This paper includes a historical analysis of the issue. In order to clarify this important topic, we tried to include papers from all around the world: Europe, North and South America, Asia, and Oceania. No African study was found. Most of these papers were written in the Northern Hemisphere. The risk factors were the same in all the papers found in our research.
Results
In table 1, the articles found in the literature review are presented in chronological order. Sixty papers published between 1944 and 2015 matched our inclusion criteria. Below, papers are presented by risk factor.

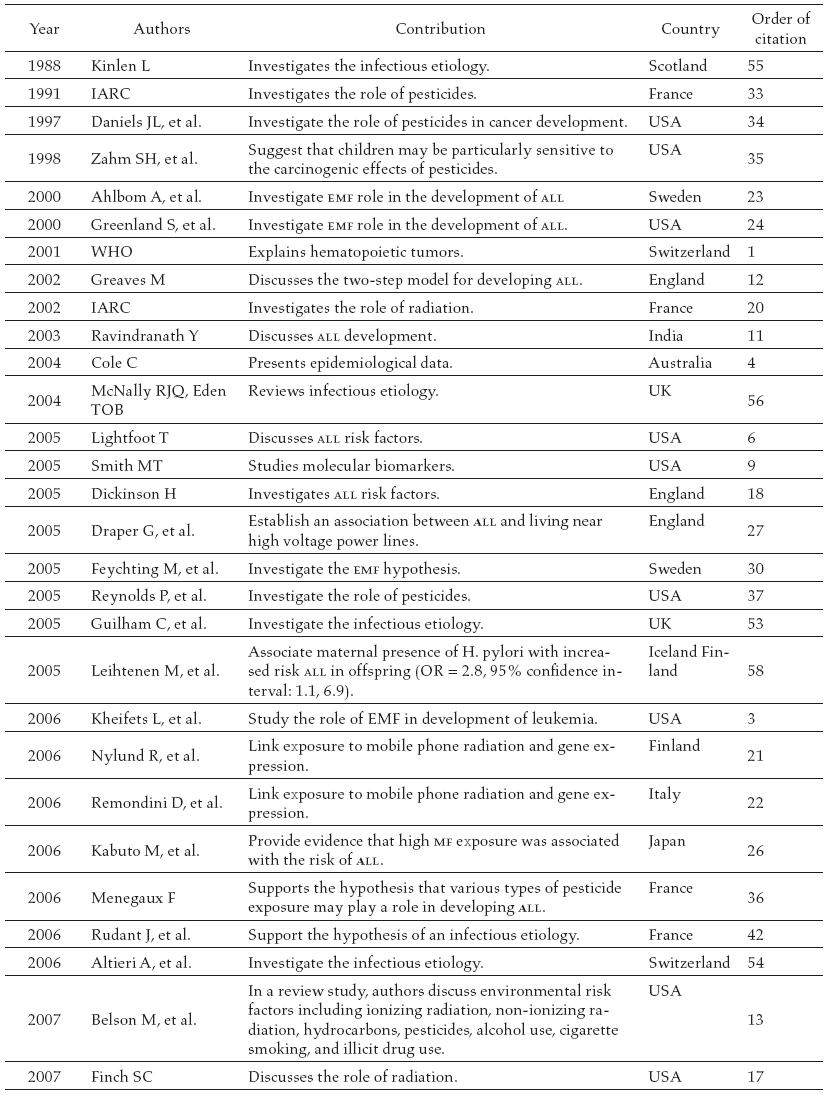
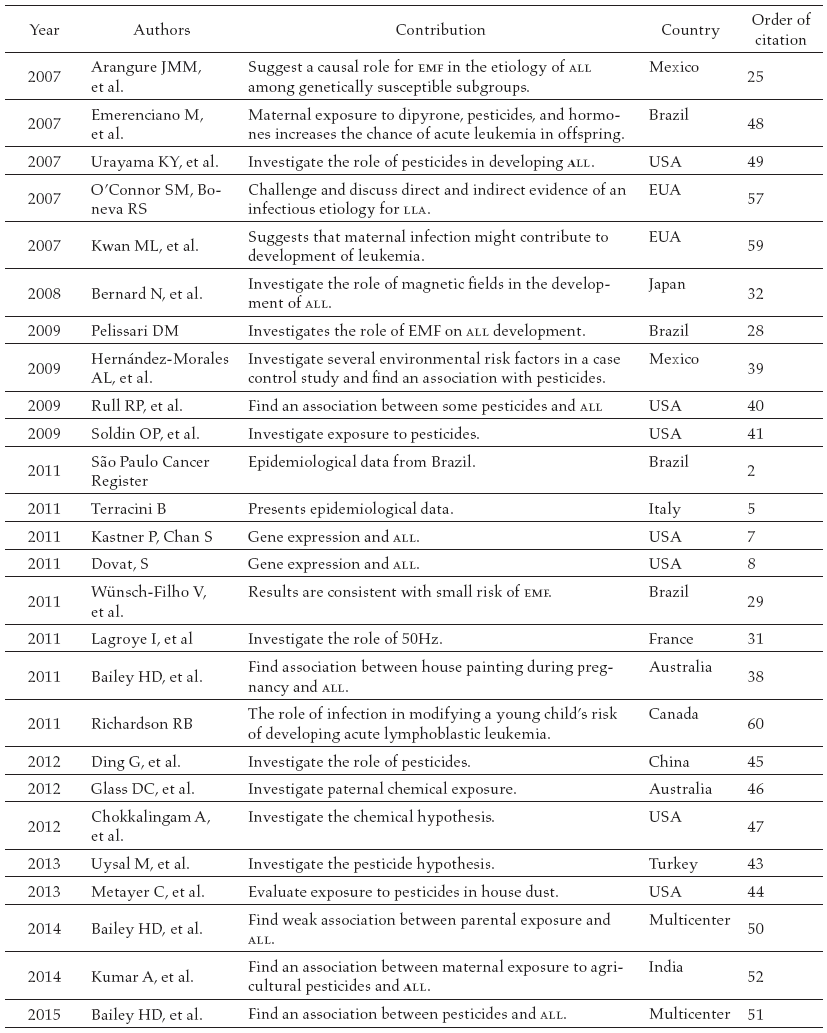
Environmental Factors and Acute Lymphoblastic Leukemia in Childhood
Ionizing radiation, benzene, HTLV-I, and Epstein-Barr virus are risk factors definitively linked to the development of some types of leukemia. The role of environmental risk factors in the development of ALL in children is unknown. Epidemiological studies of acute leukemia in children have examined possible genetic, infectious, and environmental risk factors to determine etiology. Only one environmental risk factor, ionizing radiation, has been significantly linked to ALL or AML. March and Henshaw, using epidemiological methods, sugested that ionizing radiation could induce leukemia. In 1952, Folley described an increase in the incidence of leukemia among atomic bomb survivors in Hiroshima and Nagasaki. The relationship between ionizing radiation and leukemia is established. The risk depends on the dose of radiation, the duration of exposure, and the age of the exposed person. Maternal exposure to X-rays during pregnancy increases the risk of offspring developing leukemia by 50%. The risk of preconception paternal exposure in the workplace remains uncertain (13, 14, 15, 16, 17, 18).
However, ionizing radiation does not explain the mutations that lead to childhood leukemia. There must be other risk factors that induce the genetic alterations responsible for the development of the illness. A great number of epidemiological and experimental studies had been conducted to clarify this issue but it remains uncertain. Non-ionizing radiation, pesticides, and a viral hypotheses have been seen as the most promising avenues of research.
In contrast to established evidence linking high doses of ionizing radiation with childhood cancer, research on low-dose ionizing radiation and childhood cancer has produced inconsistent results.
Non-ionizing radiation
Weithermer and Leeper found an increased risk of cancer among children living near high voltage lines in the usa; since this study, several researchers tried to link non-ionizing radiation to ALL. Power-frequency magnetic fields are classified as a possible carcinogen by the International Agency for Research on Cancer (19, 20).
There is no mechanism to explain how electromagnetic fields (EMF) could cause cancer or other health effects, although epidemiological studies found an association with high levels of exposure. Heating is the only known mechanism for the action of electromagnetic fields. However, many researchers consider the possibility of other unknown mechanisms. Human cell experiments show that electromagnetic fields can have biological effects (21, 22).
In pooled analyses of epidemiological studies, Ahlbom found that exposure to power frequency magnetic fields greater than 0.4 μT was associated with a 2-fold increase in the risk of childhood leukemia. Greenland found a 1.7-fold increase in the risk of leukemia in children living in proximity to 0,3μT (23, 24).
In a case-control study with Down Syndrome, Arangure suggested that EMF can play a role in leukemia among genetically susceptible people. In Japan, where there is high exposure to EMF, Kabuto found an association with ALL in a case-control study. Living near high voltage lines at the time of birth has a relationship to developing leukemia (25, 26, 27).
Pelissari reviews the case-control studies published on this subject. Of 152 articles tracked in different databases, ten studies from North America, Asia, and Europe met the defined selection criteria, with patients diagnosed from 1960 to 2004. The authors identify methodological limitations in these articles, including difficulties with the procedures for assessing exposure (28).
In a case-control study conducted in Brazil, the authors found an increased risk between children exposed to extremely low frequency magnetic fields equal to or greater than 0.3 microT, compared to children exposed to levels below 0.1 microT (odds ratio 1.52; 95% confidence interval 0.46-5.01) when measurements were done at night. Children living within 200m of power lines presented an increased risk of ALL (or 1.67; 95% CI 0.49-5.75), compared to children living at 600m or more from power lines. For those living within 50m of power lines the or was 3.57 (95% CI 0.41-31.44). These results are consistent with the small risks reported in other studies (29).
Acording to Feychtyng, EMF can act as a promoter or progressor, enlarging the genetic alteration caused by another agent. Lagroye suggested the use of modeling with animals to evaluate the potential for children exposed to 50-60 Hz extremely low frequency (ELF) magnetic fields (MFS) to develop leukemia, especially when the study focuses on pregnancy (30, 31).
Bernard used a rat model in which B acute lymphoblastic leukemia was chemically induced by a nitrosurea derivative. From the onset of the chemical treatment, the animals were also exposed to ELF-MFS (100 microT, sinusoidal 50 Hz MFS), with or without harmonics. The experiment was conducted on 280 rats. Body weight and survival time, percentage of bone marrow blast cells, cumulative incidence of leukemia and type of leukemia in the unexposed groups and in the groups exposed to 50 Hz MFS with and without harmonics was compared. The results showed no significant differences between exposed and unexposed rats for any of these parameters (p > 0.05) (32).
Pesticides
Chemicals are strongly associated with cancers in general and benzene is a chemical product linked to leukemia, especially when there is exposure in an occupational environment. In the case of infant ALL, pesticides showed epidemiological associations. Pesticides have a relationship to other childhood cancers and iarc considers non-arsenic pesticides a probable human carcinogen (33).
Daniels and Zahm suggest that home use of pesticides can be associated with the incidence of leukemia. In a case-control study, Menegaux analysed the use of pesticides in the home and the incidence of childhood leukemia. The authors found an association between the use of home and garden pesticides during pregnancy and childhood and an increased incidence of leukemia. Reynolds did not find any risk for all in children living near regions of intense use of agricultural pesticides in Califórnia (34, 35, 36, 37).
Bailey found evidence of a modestly increased risk of all for professional pest control treatments done during the index pregnancy, and possibly in the child’s early years. These authors used data from 388 cases and 870 frequency-matched controls to analyze the potential for the professional use of pesticides to cause leukemia and conducted a meta-analysis of their findings in relation to the findings of previously published studies. The ors for any professional pest control treatments were 1.19 (95% CI 0.83, 1.69) in the year before pregnancy, 1.30 (95% CI 0.86, 1.97) during pregnancy, and 1.24 (95% CI 0.93, 1.65) for those done after the child’s birth. The ORs for exposure after birth were highest when exposure occurred between the ages of two and three years. ors were elevated for termite treatments before birth. ors were higher for pre-B than for T cell ALL and for t (12;21) (ETV6-Runx-1) than other cytogenetic sub-types. The pooled or from a meta-analysis of their study with three previous studies of professional pest control treatments during pregnancy was 1.37 (95% ci 1.00, 1.88) (38).
A case-control study conducted in Mexico showed that the domestic or garden use of pesticides three months before pregnancy may play a role in the etiology of acute leukemia in children. Sociodemographic and gyneco-obstetric factors, perinatal features, parents’ use of alcohol or smoking, exposure to electromagnetic fields, and exposure to pesticides 3 months before and during pregnancy were assessed. Results indicated an increased OR for domestic (OR = 2.1, ci 95 = % 1.5-2.9) or garden (OR = 1.8, ci 95 % = 1.2-2.6) use of pesticides three months before pregnancy. A non-significant frequency was observed with previous fetal death, neonatal jaundice, smoking 3 months before pregnancy, father’s intake of alcohol, pesticide use during pregnancy at home or garden, and living close (< 60 m) to agricultural fields (39).
Identification of specific pesticides that may play a role in the etiology of childhood leukemia may help to clarify the role of these chemicals in the development of acute lymphoblastic leukemia (40, 41, 42).
This issue was evaluated by Rull et al. The authors categorized pesticides into groups based on similarities in toxicological effects, physicochemical properties, and target pests or uses, then conducted a case-control study using residential histories collected from the families of 213 ALL cases and 268 matched controls enrolled in the Northern California Childhood Leukemia Study. The authors assessed residential proximity within a half-mile (804.5m) of pesticide applications by linking address histories with reports of agricultural pesticide use. Elevated ALL risk was associated with lifetime moderate exposure, but not high exposure, to certain physicochemical categories of pesticides including organophosphates, chlorinated phenols, and triazines, and with pesticides classified as insecticides or fumigants (40).
Soldin et al. evaluated the associations of organophosphate exposure in a residential environment with the risk of developing of acute lymphoblastic leukemia in children. They analyzed environmental exposure by questionnaire and by urinalysis of pesticide metabolites. More case mothers (33%) than controls (14%) reported using insecticides in the home (P < 0.02), and pesticide levels were higher in cases than in controls (P < 0.05). Statistically significant differences were found between children with ALL and controls for the organophosphate metabolites diethylthiophosphate (P < 0.03) and diethyldithiophosphate (P < 0.05) (41).
A study conducted in Turkey investigated the relationship between cancer types and the total area of agricultural fields where greenhouse activities were performed. The results of this study showed that there was a statistically significant relationship between an increased incidence of multiple myeloma, acute lymphocytic leukemia, and malignant melanoma and an increase in the total area of agricultural fields (43).
Metayer examined the association between exposure to herbicides and childhood acute lymphoblastic leukemia (ALL). Dust samples were collected from homes of 269 all cases and 333 healthy controls in California, (<8 years of age at diagnosis/reference date and residing in same home since diagnosis/reference date) using a high-volume surface sampler or household vacuum bags. Amounts of agricultural or professional herbicides and residential herbicides were measured. These data suggest that levels of chlorthal and possibly alachlor in household dust are associated with increased risks of childhood ALL (44).
Pyrethroid pesticides are used in China, but few studies have focused on pyrethroid metabolites. In a case control study conducted in Shanghai between 2010 and 2011, Ding investigated five nonspecific metabolites of pyrethroid pesticides found in children’s urine and examined the correlation with childhood leukemia. The study included 176 children aged 0-14 years and 180 controls matched for age and sex. The findings suggest that urinary levels of pyrethroid metabolites may be associated with an elevated risk of childhood ALL and represent a quantitative previous exposure assessment for childhood leukemia (45).
Glass et al. conducted a case control study in Australia in which they evaluated the association between parental occupational exposure to pesticides and its influence on the development of acute lymphoblastic leukemia in offspring. Paternal occupational exposure to pesticides before or around conception was not related to increased risk of childhood ALL. There was a low prevalence of occupational exposure among women (46).
Individual xenobiotic transport and metabolism pathways may play a role in the risk of childhood ALL. Chokkalingam et al. conducted a population-based epidemiological study of 377 cases and 448 controls in California, utilizing a haplotype-based approach to evaluate 42 xenobiotic transport and metabolism genes in conjunction with data on self-reported household chemical exposures. They identified significant associations of childhood ALL risk with haplotypes of ABCB1, ARNT, CYP2C8, CYP1A2, CYP1B1, and IDH1. In addition, certain haplotypes showed significant joint effects with self-reported household chemical exposure on risk of childhood ALL. Specifically, elevated risks associated with the use of paint in the home (at any time) and indoor insecticides (pre-birth) were limited to subjects carrying specific haplotypes of CYP2C8 and ABCB1, respectively (47).
The role of household exposure to pesticides in the etiology of childhood hematopoietic malignancies was investigated by Rudant et al. They used the national registry-based case-control study escale (Etude sur les cancers de l’enfant) carried out in France over 2003-2004. Maternal household use of pesticides during pregnancy and paternal use during pregnancy or childhood were reported by mothers in a structured telephone survey (42). Insecticides (used at home, on pets, or for garden crops), herbicides, and fungicides were distinguished. Insecticide use during pregnancy was significantly associated with both lymphoblastic and myeloblastic childhood acute leukemia (OR = 2.1; 95% ci, 1.7-2.5). Paternal household use of pesticides was also related to acute leukemia (OR = 1.5; 95% ci, 1.2-1.8). The study findings strengthen the hypothesis that domestic use of pesticides may play a role in the etiology of childhood hematopoietic malignancies.
A study conducted by the Brazilian Collaborative Study Group on Infant Acute Leukemia found that Brazilian mothers exposed to pesticides had an increased chance of giving birth to babies with infant acute leukemia (OR = 2.27; 95% CI = 1.56-3.31) (48).
Individual susceptibility has been analyzed. The multidrug resistance gene (MDR1) plays an important role in protecting cells against lipophilic xenobiotics. Urayama et al. examined susceptibility associated with the presence or absence of MDR1 among children enrolled in the Northern California Childhood Leukemia Study, and predicted haplotypes and their effect modifying the association between indoor insecticide exposure and the risk of childhood acute lymphoblastic leukemia (ALL). This study suggested that children carrying the haplotype cgc may be less susceptible to the leukemogenic effects of indoor insecticide exposures (49).
More recently, Bailey et al. found a weak association between parental exposure to pesticides and ALL. Association with AML was more evident. The same group analyzed data from 12 case-control studies in the Childhood Leukemia International Consortium using logistic regression, and found an association between domestic pesticides and ALL in three periods: before conception, during pregnancy, and after birth. Odds ratios (ORS) were 1.39 (95% confidence interval [CI]: 1.25, 1.55) (using 2,785 cases and 3,635 controls), 1.43 (95% CI: 1.32, 1.54) (5,055 cases and 7,370 controls) and 1.36 (95% CI: 1.23, 1.51) (4,162 cases and 5,179 controls), respectively (Bailey et al., 2015). In an elegant case control study conducted in India, Kumar et al. found increased risk of leukemia in children whose mothers were exposed to agricultural pesticides during pregnancy (50, 51, 52).
Infectious agents
T-cell leukemia in adults is caused by a virus: human T-cell leukemia virus type 1 (HTLV-I). It is an endemic leukemia in Japan, the Caribbean, and parts of Central Africa, and its distribution is linked to the prevalence of the virus in the population (1).
Viral infection as an etiologic factor for childhood leukemia has been evaluated for the last 70 years. Kinlen assumed that an increase in the number of leukemia cases can be seen in places where there is a mixing population. Leukemia would be a rare response to a normal infeccious agent. In isolated communities, most people would not be exposed to this agent. A mixed population could lead to a new exposure and this could increase the risk of leukemia. Some researchers believe that this delay in exposure to infeccious agents in the first year of life would not prepare the immune system to respond to infections (53, 54, 55, 56, 57).
Different reseachers have investigated the possibiliy that an infectious agent could be the cause of ALL. When Greaves proposed his two-step model to explain childhood leukemia, he suggested that the second mutation could be related to an infection and that a delay in exposure to infectious agents could increase the number of preleukemic clones. This hypothesis agrees with the two-hit hypothesis proposed by Knudson in 1971 (12, 58).
Several indirect markers of exposure to infectious agents have been used to evaluate its relation to leukemia. Some of these markers, such as birth order and unprivileged environment, are inversely associated with ALL (54).
Gilham et al. tested the hypothesis that reduced exposure to common infections in the first year of life increases the risk of ALL in childhood. Social activity and day care in the first year of life were used as a parameter to evaluate the exposure of children to infections. An association has been found between social activity in first year of life and a smaller risk of developing leukemia (53).
Rudant et al. evaluated an association between the risk of leukemia at ages 0-7 years and population mixing at the birthplace. An increase of risk was found for children living in places with greater population mixing (42).
Maternal infection during pregnancy is associated with a greater risk of developing ALL in childhood. Kwan et al. analysed infectious diseases during pregnancy and the risk of ALL, and found an increase in the risk in the group of children whose mothers had influenza/pneumonia and sexually transmited diseases. Leihtenen et al. showed an association between maternal contamination with Helicobacter pylori and leukemia in offspring (56, 59, 60).
Richardson suggested that heat shock instigated by infections, particularly in infancy, stimulates Th1 pro-inflammatory cytokines and an apoptosis-inhibitory environment. This infective stress also increases the number of cooperating oncogenic mutations in pre-leukemic cells, especially if the primary adaptive immune response is delayed (61).
The viral hypothesis is considered plausible since this is a disease of cells involved in the process of immunity against pathogens and there is the possibility of an uncommon response to a common infectious agent. However, no virus has been definitively associated with childhood ALL and there are no data regarding a viral insertion in leukemical cells (54, 57).
Discussion
About our findings in this review, it is very clear that some of the most important environmental risk factors are present, such as exposure to ionizing radiation in utero or after birth, which leads us to think that radiation exposure resulting from accidents and other adverse events may play a role in this scenario, where many factors interact. Exposure to viruses may be an important event that triggers the process that leads to leukemia.
Despite the many efforts of the scientific community, the environmental factors involved in the development of childhood acute lymphoblastic leukemia remain uncertain. To clarify this issue, researchers might try the following new approaches:
Synergistic action of environmental factors should not be ruled out. It may explain why epidemiological studies found only a weak relationship between leukemia and specific risk factors.
More studies should focus on the period of pregnancy. If the first event leading to leukemia occurs in utero, the period of pregnancy must be examined. Nine months is a relatively short time and some answers may be found.
Perhaps we should consider unexamined factors such as the influence of environmental air pollution. Up to this time, few studies have considered this risk.
Finally, it is very important to design and conduct experimental research linking the genetic alteration of genes, Ikaros, for example, to risk factors that showed correlations with childhood leukemia in epidemiological studies.
The material cost of childhood all diagnosis and treatment is high. Despite advances in diagnosis, therapy, and prognosis with treatment, ALL continues to be the most significant type of childhood cancer.
Further, the cost of childhood leukemia is not only material. There is also a social and psychological burden. According to the theory proposed by Greaves based on a paper by Knudson, there are at least two moments when children are exposed to environmental factors that can lead to the development of leukemia. If the risk factors involved in the process can be identified, they can be avoided (10, 58).
In recent decades, molecular research has made a significant contribution to our understanding of the variability of individual responses to environmental carcinogens, demonstrating that some groups are more susceptible than others. With this in mind, it is necessary to identify the risk factors that can act on the development of leukemia.
As we have shown, few papers on this topic have been written in South America and none originated in Africa. These findings suggest that more efforts are necessary to study leukemia in children in Latin America. Although the results of the available papers may be applicable in Latin America, it is desirable to replicate existing studies in order to confirm their findings.
References
1. Jaffe ES, Harris NL, Stein H, Vardiman JW (eds).World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. IARC Press; Lyon: 2001. 352 p.
2. Michels FAS, Simon AS, Sconza AC, Veneziano DB, Latorre MRDO. Registro de Câncer de Sao Paulo; Sao Paulo: 2011. Cancer in Sao Paulo 1997–2008: incidence, mortality, cancer trend in Sao Paulo municipality.
3. Kheifets L, Swanson J, Greenland S. Childhood leukemia, electric and magnetic fields, and temporal trends. Bioelectromagnetics. 2006;27:545-52.
4. Cole C. Registering childhood cancers. Lancet. 2004;364(9451):2074-6.
5. Terracini B. Epidemiology of childhood cancer. Environ Health. 2011;5:10(Suppl1):88.
6. Lightfoot T. Aetiology of childhood leukemia. Bioeletromagnetics. 2005;(Supl7):S5-S11.
7. Kastner P, Chan S. Role of Ikaros in T-cell acute lymphoblastic leukemia. World J Biol Chem. 2011;2(6):108-14.
8. Dovat S. Ikaros in hematopoiesis and leukemia. World J Biol Chem. 2011 Jun;2(6):105-7.
9. Smith MT, McHalle CM, Wiemels JL, Zhang L, Wiencke JK, Zheng S, et al. Molecular biomarkers for the study of childhood leukemia. Toxicol Appl Pharmacol. 2005;206:237-45.
10. Greaves MF. Speculations on the cause of childhood acute lymphoblastic leukemia. Leukemia. 1988;2:120-5.
11. Ravindranath Y. Recent advances in pediatric acute lymphoblastic and myeloid leukemia. Curr Opin Oncol. 2003;15:23-35.
12. Greaves M. Childhood leukemia. BMJ. 2002;324:283-7.
13. Belson M, Kingsley B, Holmes A. Risk factors for acute leukemia in children: A review. Environ Health Perspects. 2007;115:138-45.
14. March HC. Leukemia in radiologists. Radiology. 1944;43:275-8.
15. Henshaw PS, Hawkins JS. Incidence of leukemia in physicians. J Nat Cancer Inst. 1944; 4:339-40.
16. Folley JH, Borges W, Yamasaki T. Incidence of leukemia in survivors of the atomic bombs in Hiroshima and Nagasaki. Am J Med. 1952;13:311-21.
17. Finch SC. Radiation-induced leukemia: Lessons from history. Best Pract Res. 2007;20(1):109-18.
18. Dickinson H. The causes of childhood leukemia. BMJ. 2005;330:1279-80.
19. Wertheimer N, Leeper E. Electrical wiring configurations and childhood cancer. Am. J. Epidemiol. 1979;109(3):272-84.
20. IARC. Non-ionizing radiation, part 1: Static and extremely low-frequency (ELF) electric and magnetic fields. [internet] Monographs on the evaluation of carcinogenic risk to humans. 2002;80. In: https://monographs.iarc.fr/ENG/Monographs/vol80/mono80.pdf
21. Nylund R, Leszczynski D. Mobile phone radiation causes changes in gene and protein expression in human endothelial cell lines and the response seems to be genome-and proteome-dependent. Proteomics. 2006;6:4769-80.
22. Remondini D, Nylund R, Reivinen J, Gannes FP, Veyret B, Lagroye I, et al. Gene expression changes in human cells after exposure to mobile phone microwaves. Proteomics. 2006;6:4745-54.
23. Ahlbom A, Day N, Feytching M, Roman E, Skinner J, Dockerty J, et al. A pooled analysis of magnetic fields and childhood leukemia. British J Cancer. 2000;83(5):692-8.
24. Greenland S, Shepard A R, Kaune WT, Poole C, Kelsh M A. A pooled analysis of magnetic fields, wire codes and childhood leukemia. Epidemiology. 2000;11(6):624-34.
25. Arangure JMM, Gutierrez AF, Saldivar MLP, Gorodezky C, Avalos AM, Guzman LR, et al. Magnetic fields and acute leukemia in children with down syndrome. Epidemiol. 2007;18(1):158-61.
26. Kabuto M, Nitta H, Yamamoto S, Yamagushi M, Akiba S, Honda Y, et al. Childhood leukemia and magnetic fields in Japan: A case-control study of childhood leukemia and residential power-frequency magnetic fields in Japan. Int J Cancer. 2006;119:643-50.
27. Draper G, Vincent T, Kroll M E, Swanson J. Childhood cancer in relation to distance from high voltage power lines in England and Wales: A case-control study. BMJ. 2005;330:1290-4.
28. Pelissari DM. Campos eletromagnéticos e leucemia linfocítica aguda em crianças residentes na Região Metropolitana de São Paulo. [dissertation]. [São Paulo]: University of São Paulo, School of Public Health; 2009. 116 p.
29. Wünsch-Filho V, Pelissari DM, Barbieri FE, Sant’anna L, de Oliveira CT, de Mata JF, et al. Exposure to magnetic fields and childhood acute lymphocytic leukemia in São Paulo, Brazil. Cancer Epidemiol. 2011 Dec;35(6):534-9.
30. Feychting M, Ahlbom A, Kheifets L. EMF and health. Annu Rev Public Health. 2005;26:165-89.
31. Lagroye I, Percherancier Y, Juutilainen J, De Gannes FP, Veyret B. ELF magnetic fields: Animal studies, mechanisms of action. Prog Biophys Mol Biol. 2011 Dec;107(3):369-73.
32. Bernard N, Alberdi AJ, Tanguy ML, Brugere H, Helissey P, Hubert C, et al. Assessing the potential leukemogenic effects of 50 Hz magnetic fields and their harmonics using an animal leukemia model. J Radiat Res (Tokyo). 2008 Nov; 49(6):565-77.
33. IARC. Ocupational exposures ln insecticide application, and some pesticides. [internet] Monographs on the evaluation of carcinogenic risk to humans. 1991;53. In: http://monographs.iarc.fr/ENG/Monographs/vol53/mono53.pdf
34. Daniels JL, Olshan A F, Savitz D A. Pesticides and childhood cancers. Environ Health Perspects. 1997;105:1068-77.
35. Zahm S H, Ward M H. Pesticides and childhood cancer. Environ Health Perspects. 1998; 106(Suppl3):893-908.
36. Menegaux F, Baruchel A, Bertrand Y, Lescoeur B, Leverger G, Nelken B, et al. Household exposure to pesticides and risk of childhood acute leukemia. Occup Environ Med. 2006;63:131-4.
37. Reynolds P, Behrens J V, Günter R, Goldberg D E, Hertz A. Agricultural pesticides and lymphoproliferative childhood cancer in California. Scand J Work Environ Health. 2005;31(Suppl1):46-54.
38. Bailey HD, Milne E, de Klerk NH, Fritschi L, Attia J, Cole C, et al. Exposure to house painting and the use of floor treatments and the risk of childhood acute lymphoblastic leukemia. Int J Cancer. 2011;128(10):2405-14.
39. Hernández-Morales AL, Zonana-Nacach A, Zaragoza-Sandoval VM. Associated risk factors in acute leukemia in children. A cases and controls study. Rev Med Inst Mex Seguro Soc. 2009 Sep- Oct;47(5):497-503.
40. Rull RP, Gunier R, Von Behren J, Hertz A, Crouse V, Buffler PA, Reynolds P. Residential proximity to agricultural pesticide applications and childhood acute lymphoblastic leukemia. Environ Res. 2009 Oct;109(7):891-9.
41. Soldin OP, Nsouli-Maktabi H, Genkinger JM, Loffredo CA, Ortega-Garcia JA, Colantino D, et al. Pediatric acute lymphoblastic leukemia and exposure to pesticides. Ther Drug Monit. 2009 Aug;31(4):495-501.
42. Rudant J, Bacaine B, Ripert M, Goubin A, Bellec S, Hemon D, et al. Population-mixing at the place of residence at the time of birth and incidence of childhood leukemia in France. Eur J Cancer. 2006;42:927-33.
43. Uysal M, Bozcuk H, Karakilinc H, Goksu S, Tatli AM, Gunduz S, et al. Pesticides and cancer: The first incidence study conducted in Turkey. J Environ Pathol Toxicol Oncol. 2013; 32(3):245-9.
44. Metayer C, Colt JS, Buffler PA, Reed HD, Selvin S, Crouse V, et al. Exposure to herbicides in house dust and risk of childhood acute lymphoblastic leukemia. J Expo Sci Environ Epidemiol. 2013 Jul; 23(4):363-70.
45. Ding G, Shi R, Gao Y, Zhang Y, Kamijima M, Sakai K, et al. Pyrethroid pesticide exposure and risk of childhood acute lymphocytic leukemia in Shanghai. Environ Sci Technol. 2012 Dec 18;46(24):13480-7.
46. Glass DC, Reid A, Bailey HD, Milne E, Fritschi L. Risk of childhood acute lymphoblastic leukemia following parental occupational exposure to pesticides. Occup Environ Med. 2012 Nov; 69(11):846-9.
47. Chokkalingam A, Metayer C, Scelo GA, Chang JS, Urayama KY, Aldrich MC, et al. Variation in xenobiotic transport and metabolism genes, household chemical exposures, and risk of childhood acute lymphoblastic leukemia. Cancer Causes Control. 2012 Aug;23(8):1367-75.
48. Emerenciano M, Koifman S, Pombo-de-Oliveira MS. Acute leukemia in early childhood. Braz J Med Biol Res. 2007 Jun;40(6):749-60.
49. Urayama KY, Wiencke JK, Buffler PA, Chokkalingam AP, Metayer C, Wiemels JL. MDR1 gene variants, indoor insecticide exposure, and the risk of childhood acute lymphoblastic leukemia. Cancer Epidemiol Biomarkers Prev. 2007 Jun;16(6):1172-7.
50. Bailey HD, Fritschi L, Infante-Rivard C, Glass DC, Miligi L, Dockerty JD, et al. Parental occupational pesticide exposure and the risk of childhood leukemia in the offspring: Findings from the childhood leukemia international consortium. Int J Cancer. 2014 Nov 1;135(9):2157-72. Doi: 10.1002/ijc.28854
51. Bailey HD, Infante-Rivard C, Metayer C, Clavel J, Lightfoot T, Kaatsch P, et al. Home pesticide exposures and risk of childhood leukemia: Findings from the Childhood Leukemia International Consortium. Int J Cancer. 2015 Dec 1;137(11):2644-63. Doi: 10.1002/ijc.29631
52. Kumar A, Vashist M, Rathee R. Maternal factors and risk of childhood leukemia. Asian Pac J Cancer Prev. 2014;15(2):781-4.
53. Gilham C, Peto J, Simpson J, Roman E, Éden TOB, Greaves MF, et al. Day care in infancy and risk of childhood acute lymphoblastic leukaemia: Findings from the UK case-control study. BMJ. 2005; 330:1294-300.
54. Altieri A, Castro F, Bermejo JL, Hemminki K. Number of siblings and the risk of lymphoma, leukemia, and myeloma by histopathology. Cancer Epidemiol. Biomarkers Prev.2006;15(7):1281-6.
55. Kinlen L. Evidence for an infective cause of childhood leukaemia: Comparison of a Scottish new town with nuclear reprocessing sites in Britain. Lancet. 1988 Dec 10;2(8624):1323-7.
56. McNally RJQ, Eden TOB. An infectious aetiology for childhood acute leukaemia: A review of the evidence. BJH. 2004;127:243-63.
57. O’Connor SM, Boneva R S. Infectious etiologies for childhood leukemia: Plausibility and challenges to proof. Environ Health Perspect. 2007;115:146-50.
58. Knudson A. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA. 1971;68(4):820–3.
59. Leihtenen M, Ogmundsdottir HM, Bloigu A, Hakulinen T, Hemminki E, Gudnadottir M, et al. Associations between three types of maternal bacterial infection and risk of leukemia in the offspring. Am J Epidemiol. 2005;162(7):662-7.
60. Kwan ML, Metayer C, Crouse V, Buffler PA. Maternal illness and drug/medication use during the period surrounding pregnancy and risk of childhood leukemia among offspring. Am J Epidemiol. 2007; 165(1):27-35.
61. Richardson RB. Promotional etiology for common childhood acute lymphoblastic leukemia: The infective lymphoid recovery hypothesis. Leuk Res. 2011;11:1425-31.
Author notes
1
PhD
2
PhD
3
PhD
4
PhD
facan@usp.br



